
“Risk management is painful—not a natural act for humans to perform.” – Gentry Lee, the chief systems engineer at Jet Propulsion Laboratory (JPL).
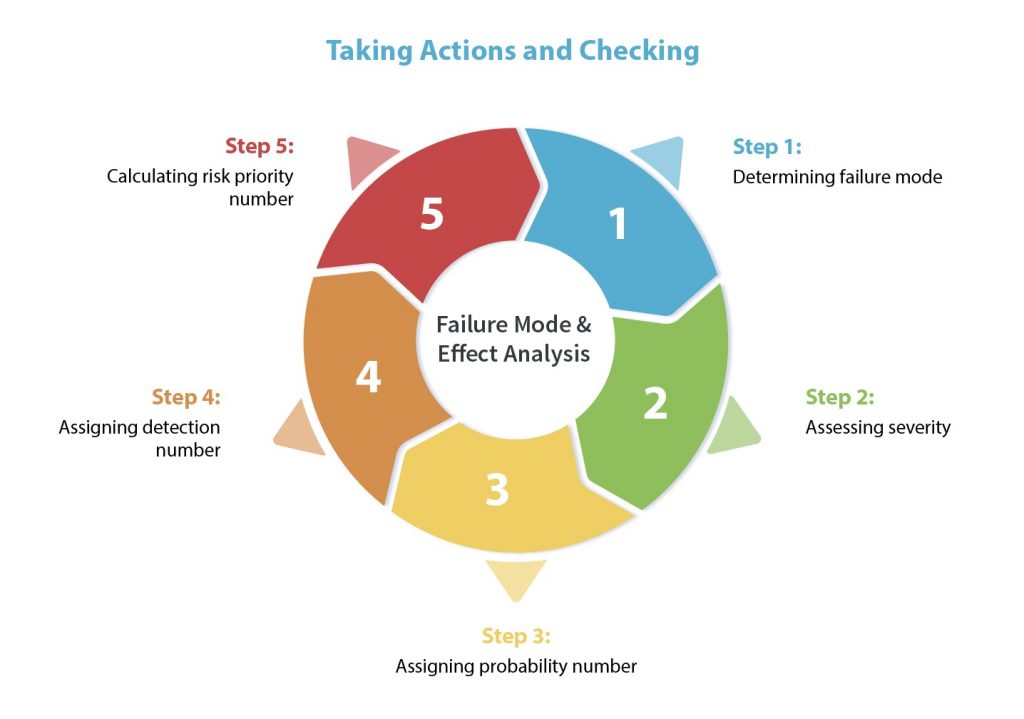
Failure Mode and Effects Analysis (FMEA) is a structured approach for identifying all possible failures in a design, manufacturing process, or a product/service. A common risk analysis tool for identifying and classifying potential failure modes based upon their severity, likelihood of occurrence and detection ratings, providing organizations with an overall Risk Priority Number (RPN) to better understand their risk profile and risk tolerance levels.
(RPN = S x L x D)
There have been multiple high-profile product failures resulting from poorly designed products or manufacturing processes. Many of these failures are still being debated heavily by consumers, with OEMs, service providers and suppliers finding themselves constantly defending their abilities to provide safe and high quality products. Examples include the recent Boeing 737 Max crash, resulting in the aviation authorities having to ground the 737 Max series, or the failure of nuclear reactor 4 at Chernobyl Nuclear Power Plant which made it one of the most contaminated locations in the world. Another example could be the failure of Takata airbags, some calling the case “the largest and most complex safety recall in U.S. history”.
FMEA isn’t just limited to major disasters, FMEA can be used to prevent all types of failure. For example, when British Airways’ key data centers lost power, 800 flights were canceled with estimates of around 75,000 travelers affected (£100 million) or Intel’s problem with the Pentium’s inability to perform math correctly ($475 Million).
Understanding FMEA
A failure mode is one possible way a system can fail due to an identifiable cause or causes. Effects are what these failures may lead to, such as product defects or dangerous outcomes for consumers. FMEA is designed to identify, prioritize and limit all these failure modes. It is important to recognize that FMEA is not a replacement for good manufacturing practices, it aims to enhance the product or process quality, combining a range of knowledge and analysis techniques to review the failure mode and assess its overall risk to the organization.
There are two broad categories of FMEA:
Design FMEA:
Design FMEAs (DFMEA) enables organizations to assess and address potential failure modes are the early or end stage of product design. DFMEA explores the possibility of malfunctions, reduced product life and safety or regulatory concerns.
Process FMEA
Process FMEAs (PFMEA) are used to analyze and maintain process control objectives. Essentially, PFMEAs are performed on a process as opposed to a product as with DFMEAs. PFMEA discovers failure that impacts product quality, reduced reliability of the process, customer dissatisfaction, and safety or environmental hazards.
Why use FMEA?
The sooner any form of failure can be discovered, the easier it will be to manage, contain & control; product recall costs will also be significantly less. If a failure is discovered late in the process, the impact can be devastating. FMEA is one of the many tools used to discover failure at its earliest and more crucial point in the product or process design phase.
When conducting FMEA organizations must consider the following:
–Ensuring all stakeholders are involved in the process: FMEA requires a full team to make it as effective as possible. If any problems arise, the dedicated risk management team can help manage issues and present solutions to mitigate critical risks.
–Having the right tool to understand the scope of FMEA: Usually, organizations have a large amount of data surrounding their risk, compliance & quality programs. Having the right tool that connects these programs is crucial. An organization’s risk ratings should be tied with work programs allowing for fully comprehensive risk analysis and mitigation.
–Documenting all processes: Organizations adhering to certain regulations or standards know how important it is to have the ability to go back and review previous documentation. If conducted right, an organization’s FMEA tool will demonstrate a commitment to meeting specific compliance & risk targets.

When to Perform FMEA
There are multiple scenarios when it makes sense to perform a Failure and Effects Analysis:
-When you are designing a new product, process or service
-When you are planning on performing an existing process in a different way
-When you have a quality improvement goal for a specific process
-When you need to understand and improve the failures of a process
It is also recommended that organizations perform an FMEA across the lifecycle of the product or process. Quality and reliability must be consistently examined for continuous improvement.
An organization’s risk profile is an extremely valuable data point. It can be used as a reference when it comes to understanding how the organization is managing its critical risk. Risk assessments give organizations the ability to associate value and meaning to their risks, which determines the level of tolerance over time. When an organization is equipped with the ability to measure risk in a way that is relative to tolerance, they can prepare for, mitigate and control their risk rating before it falls out of tolerance.
AuditComply’s risk assessment feature is developed to replicate the flow of FMEA, providing a step-by-step approach for identifying all possible failures in design or process. Organizations can prioritize according to the severity of risks, their likelihood and their detection rating.
The purpose of AuditComply’s risk assessment is to take actions that eliminate or reduce failures, starting with the highest priority one. AuditComply also documents current knowledge and actions about the risks of failure, linking all aspects of the organization’s risk, compliance & quality programs for continuous improvement.
If you would like to avail of an AuditComply risk assessment demo please contact us below.

