
GMP (Good Manufacturing Practice) audits are evaluations of the production processes and facilities used in the food and beverage industry. They assess the overall cleanliness, organization, and management of the production environment, as well as the quality control procedures used to ensure the safety and quality of food products.
GMP audits are performed to ensure that the food and beverage products produced in a facility are in compliance with regulations and standards set by government agencies such as the FDA (Food and Drug Administration) in the US and other equivalent organizations around the world. The goal of a GMP audit is to verify that the facility’s food safety and quality systems are robust and effective, and that the food and beverage products produced are safe for consumers. The audit process involves a thorough examination of the facility’s operations, including a review of policies and procedures, sanitation practices, employee training programs, and product testing methods.
The auditor will observe the production process and inspect equipment, storage facilities, and other areas to ensure that they meet the appropriate standards. GMP audits are a critical component of quality assurance and risk management in the food and beverage industry, and they play a key role in helping organizations meet regulatory requirements, maintain product quality, and protect consumers.
GMPs allow you to be competitive in the marketplace, solidifying a great reputation of trust, as well as limiting losses due to incorrect storage, recalls, or unsatisfied customers. A GMP audit will focus on a number of key areas in your production facility. Areas might include:
– Building and facilities: Inspection of the construction, layout, and design of the building and facilities to ensure they meet food safety and hygiene standards.
– Personnel practices: Examination of the training, hygiene, and behavior of employees and assess their understanding of food safety and quality procedures.
– Equipment Maintenance: Evaluation of the condition, design, and maintenance of production equipment, including the provision of appropriate tools and utensils, to ensure food safety and quality.
– Chemical management: Assessment of controls over the use of chemicals in their production processes, including the selection, handling, storage, and disposal of these substances.
– Waste management: Assessment of the procedures and facilities used for collecting, storing, and disposing of waste, including the segregation of different types of waste.
– Quality Control: Assessment of the quality control procedures used to ensure that the food and beverage products produced meet the required standards, regulation and specifications (FSMA, SQF, HACCP or BRC).
– Sanitation: Assessment of the cleanliness and maintenance of the facility, including equipment and utensils, as well as the effectiveness of pest control measures
Implementing GMPs requires a lot of groundwork, much of which is essential in the success of the established practices. It is imperative that all members of the business are competent in the expectations set in place, and thus you must develop an effective program of training, reporting and documenting.
Audits should be conducted as a means of identifying any areas within the organization where GMP standards may have slipped. Considering the complexity of GMPs, you may be wondering how best to handle the administrative strain that comes. If your process is manual, pen and paper based you may have poor insight into real performance. Managing paper-records and spreadsheets is not only time consuming, but often inconsistencies prevail due to disconnected teams and scattered data-sources.
Introducing GMP Auditing Software

In recent years, there has been a growing trend towards introducing a platform to manage GMP audits. The use of digital tools and software has many benefits:
– Efficiency: Digitizing GMP audits can greatly improve the efficiency of the audit process. With digital tools, auditors can collect, store, and analyze data in real-time, allowing for a more streamlined and organized audit process. This can help to reduce the time and effort required to complete audits, allowing organizations to focus their resources on other important areas of their business.
– Accuracy: Digitizing GMP audits also helps to improve accuracy. With digital tools, auditors can collect data using standardized forms and templates, reducing the risk of errors and omissions. Additionally, digital tools can help to ensure that audits are conducted consistently and that data is collected in a standardized manner, improving the reliability and comparability of audit results.
– Cost Savings: Digitizing GMP audits can also help organizations to reduce costs. By reducing the time and effort required to complete audits, organizations can reduce the need for additional resources, including staff time and travel expenses. Additionally, the use of digital tools can help to reduce paper-based expenses, such as printing, storage, and shipping costs.
– Improved Compliance: Digitizing GMP audits can also help organizations to improve their overall compliance efforts. With digital tools, organizations can easily track and analyze audit results, identify areas for improvement, and implement necessary changes to their production processes. This can help to ensure that organizations remain in compliance with GMP standards and regulatory requirements and that their products remain safe, high-quality, and effective.
AuditComply – Built by Auditors for Auditors
AuditComply is empowering food organizations worldwide, protecting revenue & reputation, driving compliance, reducing risk and improving yield. A mobile Audit Management solution that simplifies the process of configuring, scheduling and carrying out GMP audits. When choosing a digital GMP audit platform, it is important to consider the following capabilities to ensure your requirements are met:
1. Customizable Audit Forms – Build, replicate and optimize custom templates specific to your internal processes or regulatory standards and specify the types of data that you want collected. Create a tailored flow of subset questions to log observations, including multiple data capture elements (eg. Calculations, Thresholds, Photographic Evidence).
2. Real-Time Mobile Data Collection – Collect data in real-time, using tablets or mobile devices and synchronize it with a centralized database. This will help ensure that data is collected consistently and accurately. Conduct assessments anywhere at any time on iOS or Android and streamline evidence collection with offline mobile data capture.
3. Document Control – A centralized repository for storing, organizing and managing all documents. This includes the ability to search, retrieve and share documents as needed. Such documents may include SOPs, training materials, manuals, documentation, work instructions and more.
4. Data Anlytics & Reporting – Experience robust data analytics and reporting capabilities, allowing you to easily analyze and understand audit results. This includes the ability to generate real-time reports and dashboards to track progress and identify areas for improvement.
5. Workflow & Automation – Streamlined workflow process for review, approval and distribution of documentation, ensuring documents are updating in a timely and consistent manner.
6. Audit Trail – An audit trail of all document related activities, including who made the changes, when they were made and what changes were made. This helps ensure accountability and supports continuous improvement efforts.
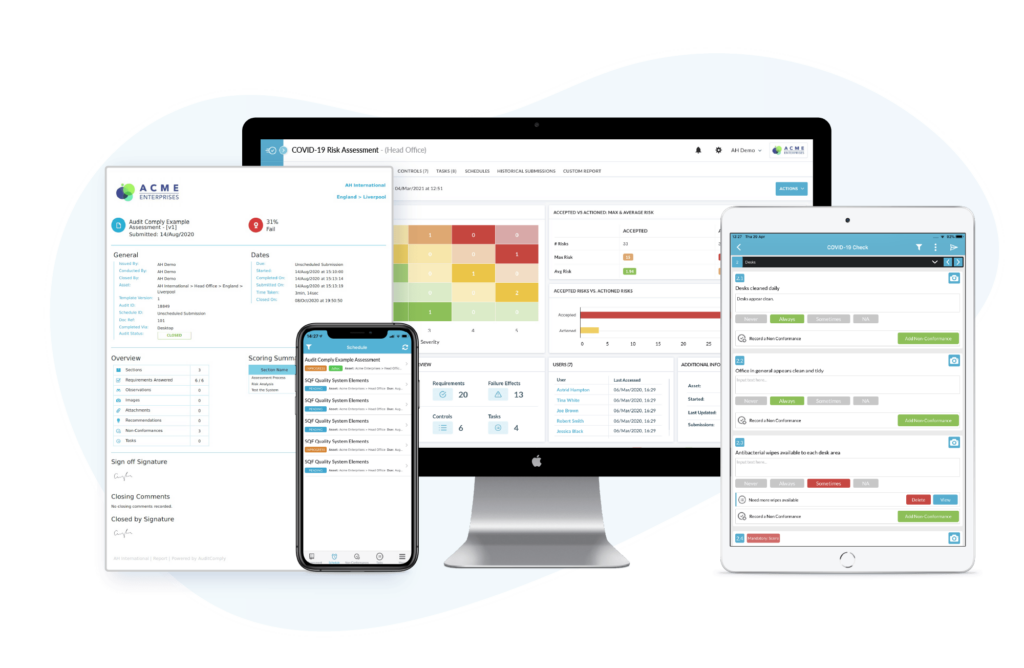
In conclusion, introducing a GMP Audit Management platform is a critical first step for organizations looking to improve their GMP compliance efforts. The use of digital tools and software can help to increase efficiency, accuracy, and cost savings, while also improving overall compliance efforts. By digitizing GMP audits, food organizations can stay ahead of the curve and ensure that their production processes meet GMP standards and regulatory requirements.
To learn more about how AuditComply can guide & evolve your GMP Management strategy, request a demo here.
